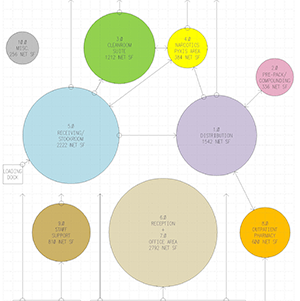
What is USP 795?
USP 795 is an important publication issued by the United States Pharmacopeia. Widely agreed to improve the safety of the compounding environment and the products produced in that environment, USP 795 is mandated by some state boards of pharmacy and recommended by others. Sterile compounding environments designed, built, and operated in compliance with USP 795 are considered by most pharmaceutical professionals as state-of-the-art.
However, its requirements also extend to architectural and environmental areas. Consequently, hospital design, construction and operations professionals should also become familiar with it.
Far-reaching regulation
USP 795 is a far-reaching regulation that governs a wide range of pharmacy policies and procedures. It is designed both to cut down on infections transmitted to patients through pharmaceutical products and to better protect staff working in pharmacies in the course of their exposure to pharmaceuticals.
Issued by U.S. Pharmacopoeia (USP), the regulation governs any pharmacy that prepares “compounded sterile preparations” (CSPs). Many pharmacies fit this description. Moreover, many large hospitals have several pharmacies–a main one and several satellite pharmacies–that will be affected.